The purpose of OLAC’s quality assurance program is to maintain consistency of animal research while ensuring compliance with our policies as well as requirements set by the Guide for Care and Use of Laboratory Animals and the USDA. The quality assurance services provided by OLAC include Animal Health Monitoring, Microbiological Monitoring, Autoclave Validation and Enrichment Program.
Animal Health Monitoring
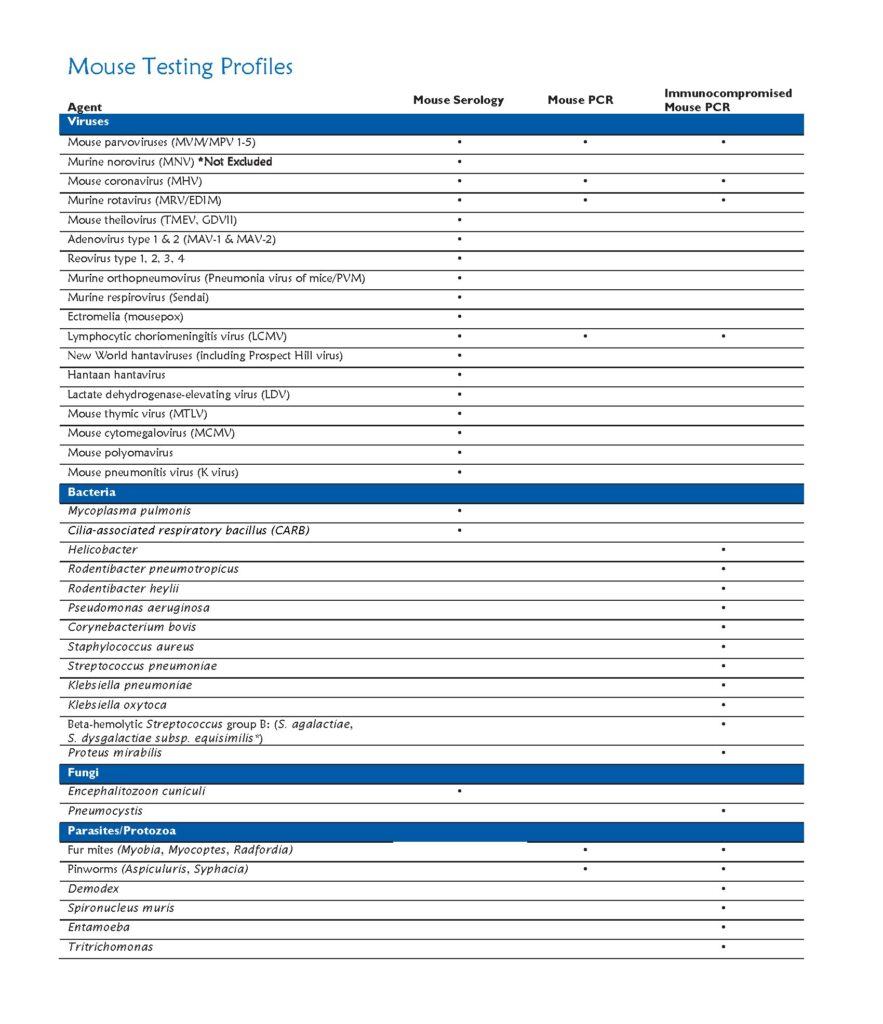
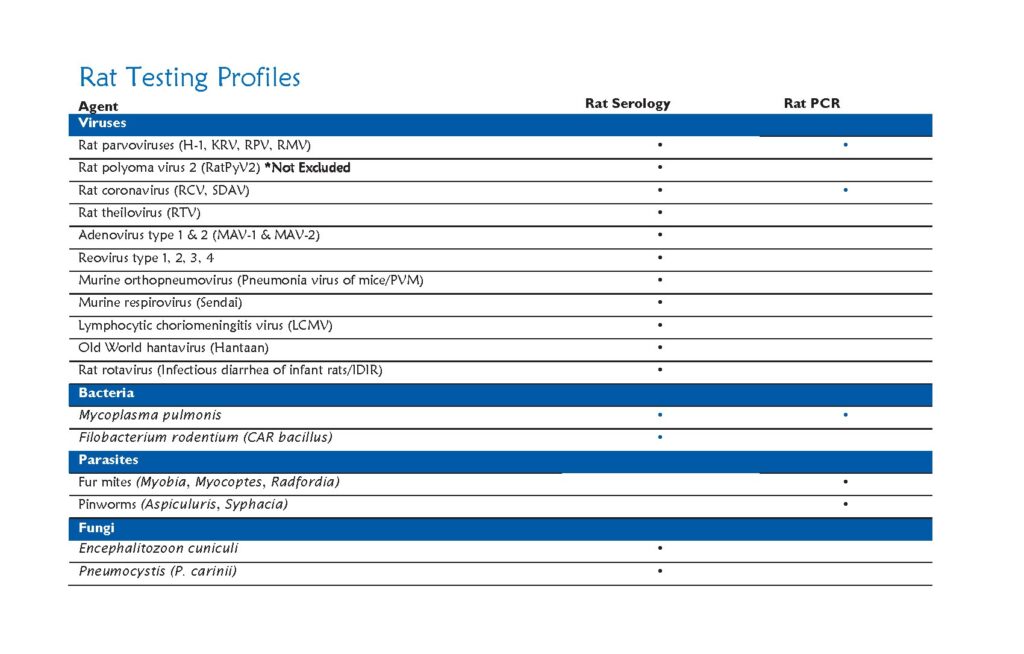
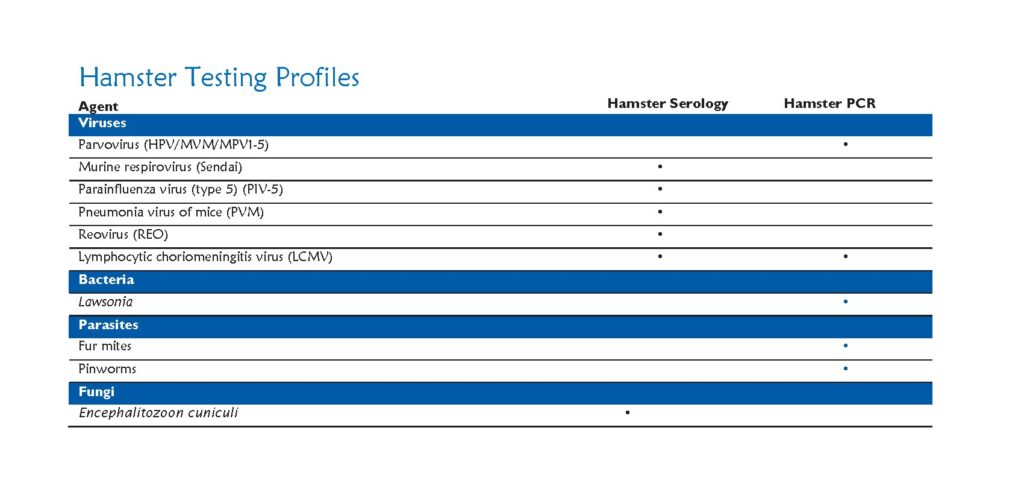
Research results derived from animal models may be altered by unwanted pathogens. Identification of pathogens in our research colonies allows early action to contain disease, thus preventing spread of disease to other colonies on campus. The health monitoring program is essential for the maintenance of rodents that are free of infectious diseases that can adversely affect research through morbidity, mortality, and by impairing physiologic and immunologic function.
For rodent populations, dirty bedding sentinels are tested every 3 months for the most common infectious agents with annual comprehensive screens for all excluded infectious agents.
Testing includes:
Serologic and PCR evaluation for infectious diseases
Parasitology: Endoparasites and ectoparasites
Necropsy for gross lesions
Microbiological Monitoring
The purpose of microbiological monitoring is to measure the efficacy of sanitation procedures in the laboratory animal facilities and PI maintained laboratories. Proper sanitation is important to any program of animal care. The Animal Welfare Act and the Guide for Care and Use of Laboratory Animals include provisions for appropriate sanitation of animal housing areas and equipment. In order to accomplish this objective, OLAC employs twice yearly microbiological monitoring as a direct way to test sanitation efforts.
Autoclave Validation Program
The laboratory animal facilities and principal investigators use autoclaves to sterilize waste and sterilize materials associated with biomedical research. The autoclave validation program performs challenge testing quarterly on all autoclaves used within UTK Laboratory Animal Facilities and on autoclaves used to sterilize surgical instruments. Validation tests are performed using a biological indicator containing a standard challenge population of Geobacillus stearothermophilus spores that are naturally resistant to high heat and pressure. Inactivation of these spores can be achieved with a 16 minute exposure to typical autoclave conditions of 121C and 15psi. Biological indicators can be obtained from OLAC free of charge.
Enrichment Program
The enrichment program is designed to enhance the life of all of our laboratory animals. With the oversight of the Attending Veterinarian, the OLAC Enrichment Coordinator develops the enrichment program to provide for the specific needs for each of our laboratory animal species. The enrichment program provides for species specific behavior ranging from play time for dogs and cats to providing foraging items for mice and rats.